About - Publications - Blog
Empirical study of the characteristics of organic solar cells
We compare the reported characteristics of 36 different bulk heterojunction organic solar cells fabricated and characterized by different research institutes. By comparing our results with our similar empirical study from 2006, we chart the technological progress for organic solar cells. Among others, we find an empirical threshold of 0.3 eV between the LUMOs of donor and acceptor, necessary for exciton dissociation. This work was performed by one of my students Tom Vandecaveye as part of his (excellent) master thesis, titled "A simulation program for and empirical study of photovoltaic organic multi-junction solar cells".
1. Introduction
In 2006, we compared the reported characteristics of 22 different bulk heterojunction
organic solar cells fabricated and characterized by different research institutes
[1]. Now (begin 2014), we repeat this study but now for the organic solar cells
fabricated today.
More specifically:
- We compare the improvements of the characteristics between the 2006 study [1] and the current state-of-the-art.
- In the 2006 study, we concluded that the current was the bottleneck for
efficiency improvements for organic solar cells. We analyze whether this is
still the case.
Table 1. The efficiency and year of publication of the 36 organic solar cells considered in this study. The second-to-last column refers to the considered photovoltaic cell. The last column refers to the energy levels of the materials.
- We determine an empirical threshold between the energy levels necessary for exciton dissociation.
- We search for a relationship between the interface bandgap and the open circuit voltage.
- We study the relation between the voltage factor and open circuit voltage for the organic solar cells.
- We compare the photovoltaic cells with the two most frequently used acceptors.
For this empirical research, we studied and compared the reported characteristics of 36 different organic solar cells fabricated and characterized by different research institutes (Table 1). We only considered solar cells where both the donor and the acceptor are organic solid state materials. Organic-inorganic hybrid cells, liquid dye sensitized solar cells and multi-junction cells were omitted for this study. For each donor-acceptor material combination, only the cell with the highest reported efficiency was taken into account. The cells were characterized under illumination with the standard A.M. 1.5 spectrum and an intensity of 100 mW/cm². Two cells [23] were characterized with a lower intensity (95 mW/cm²) and one [4] at a higher intensity (128 mW/m²). We adapted the Jsc linearly of those cells to compare them with the cells characterized at 100 mW/cm². No corrections were made for the Voc or FF.
This resulted in experimental data of 36 bulk heterojunction organic solar cells, published within the time frame 2009 - 2013. In a bulk heterojunction solar cell, the active layer consists of a nanoporous interpenetrating network of the donor and the acceptor. As expected, no bilayer cells were found with a higher efficiency as their bulk heterojunction counterpart. All 36 cells use as transparent hole-contact Indium Tin Oxide (ITO) cathode. As electron contact, different materials can be used. Al is used in most cases (26 cells), Ag is used in 9 cells [7,8,15,30,32].
The highest standard efficiency published for organic solar cells is at this time 9.214 % for an ITO / PFN interlayer / PTB7:PC71BM / MoO3 / Al – cell [36]. This cell has an active area of 8 mm², and is characterized at 100 mW/cm² by an open circuit voltage Voc of 0.754 V, a short circuit current density Jsc of 17.46 mA/cm², and a fill factor FF of 69.99 %. We want to remark that the world record efficiency reported for an organic solar cell is 12.0 %, fabricated by the company Heliatek. But since they don’t disclose the used materials, we could not include this cell in this study.
2. Results
The power conversion efficiency η of a solar cell is given by the formula:

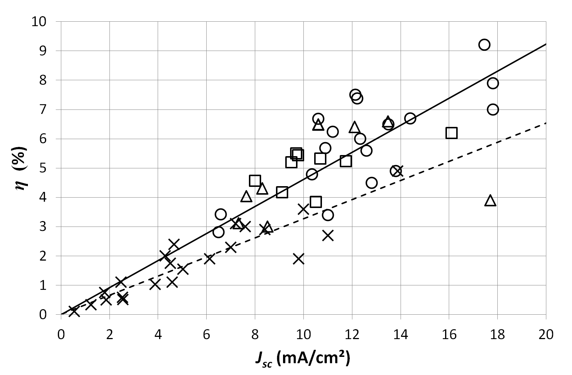
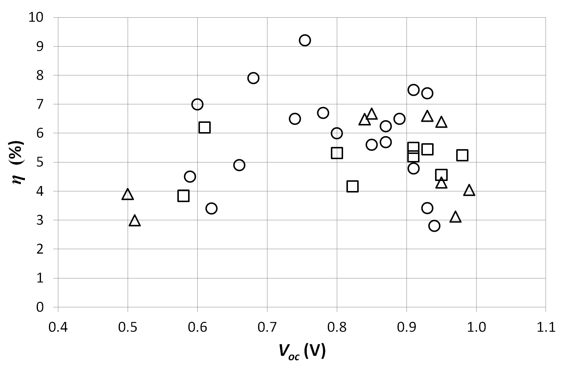
where Voc is the open circuit voltage, Jsc the short circuit current density, FF the fill factor and Pin the incident solar light power. When we plot the measured characteristics of the different cells versus the efficiency (Figure 1 and 2), we see a clear linear correlation for the short circuit current density Jsc (and to a lesser extend for the fill factor FF). An increase of 1 mA/cm² results roughly in an efficiency gain of 1/2 of a percent absolute. In our 2006 study, this was 1/3 of a percent absolute. However, this linear correlation is not seen for the efficiency versus the open circuit voltage Voc (Figure 3). This empirical result indicates that, with the current state of the technology, the current is the limiting factor for reaching higher efficiencies, and not the Voc. The average Voc of the cells (0.81 V) is already satisfyingly high. Where there is no clear relationship between the Voc and the efficiency, this relation is present for the Jsc. Of course, reaching higher voltages will increase the efficiency of the solar cells, but a higher efficiency gain can be obtained by focusing on reaching higher currents, which is an important bottleneck for the breakthrough of organic solar cells. These conclusions are the same as our 2006 study. Also then, the Voc was already satisfyingly high (with an average of 0.75 V). The average Voc hardly increased (+ 8 % relative) whereas the average Jsc more than doubled (from 5.5 to 11.5 mA/cm²). Figure 1 indicates the progress during the last 8 years in Jsc. This proves the conclusion from the 2006 study that the current (and not the voltage) was (and still is) the limiting factor. Also the average fill factor FF increased from 45 % in 2006 to 59 % now (a relative increase of 31 %).
Figure 1. The efficiency η of the studied solar cells versus the short circuit current density Jsc. The crosses represent the cells from the 2006 study. The circles and squares represent respectively cells from the current study where PC70BM and PC60BM are the acceptor material. The triangles represent cells with other acceptor materials. The straight line is the linear fit through the origin for the cells of the current study. The dashed line is the linear fit through the origin for the 2006 study.
The absorption of a photon leads to the creation of an exciton in the donor
of a bulk heterojunction solar cell. For the charge carriers to contribute to
the current, the exciton needs to be dissociated into an electron and a hole
before recombination occurs. The exciton is dissociated at the discontinuous
potential drop at the interface between donor and acceptor. The electron is
transferred to the electron acceptor due to its high electron affinity.

Figure 2. The efficiency η of the studied solar cells versus the fill factor FF. The circles and squares represent respectively cells where PC70BM and PC60BM are the acceptor material. The triangles represent cells with other acceptor materials. The straight line is the linear fit through the origin.
Figure 3. The efficiency η of the studied solar cells versus the open circuit voltage Voc. The circles and squares represent respectively cells where PC70BM and PC60BM are the acceptor material. The triangles represent cells with other acceptor materials.
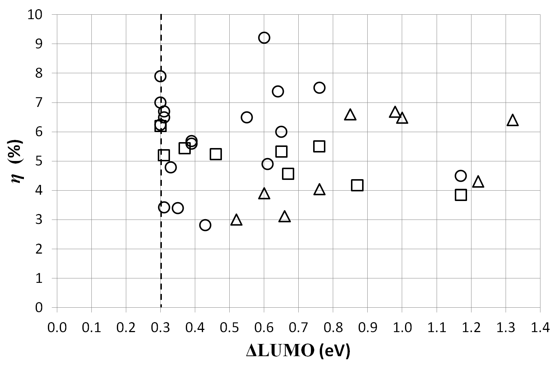
Figure 4. The efficiency η of the studied solar cells versus the energy difference between the LUMOs of donor and acceptor. The circles and squares represent respectively cells where PC70BM and PC60BM are the acceptor material. The triangles represent cells with other acceptor materials. The dashed line represents the empirical threshold of 0.3 eV.
A necessary condition for exciton dissociation is that the difference between the LUMOs of the donor and acceptor (ΔLUMO) is higher than the exciton binding energy. Although the value of the exciton binding energy in different materials is often a subject of discussion, values between 0.1 eV and 2 eV are published [37]. Figure 4 shows the efficiency as a function of the energy difference between the LUMOs of the donor and the acceptor of the studied solar cells. Most energy differences are lying between 0.3 eV and 0.8 eV, although some even reach 1 eV and higher. If ΔLUMO is higher than necessary for the exciton dissociation, the electron loses useful energy that can’t contribute any more to the output power, although this is not clear from Figure 4. An empirical threshold of 0.3 eV is found between the LUMOs of the donor and acceptor. Because no working cells were found below that threshold, a minimal difference of 0.3 eV between the LUMOs can be considered as necessary for exciton dissociation. This is in accordance with the threshold found by others [38,39] and 0.1 eV higher than the 2006 study. A possible explanation for the 0.1 eV discrepancy between the current and the 2006 study could be the error margin in determining the energy levels of organic materials [40,41].
The distance between the HOMO of the donor and the LUMO of the acceptor is
considered as the thermodynamic limitation of the useful energy [42]. We call
this value the interface bandgap Ei. For an organic solar cell with ohmic contacts,
the open circuit voltage Voc is linearly dependent on the interface bandgap
Ei. This linear relationship was proven for the variation of the HOMO level
of the donor [38,43,44] and of the LUMO-level of the acceptor [45-47]. For a
cell with non-ohmic contacts, the Voc is dependent on the work function difference
of the electrodes [48].
Figure 5 shows the Voc as a function of the interface bandgap Ei for the studied
solar cells. We would expect a linear relationship, but as in the 2006 study,
this relationship can not be found. Possible explanations could be the influence
of bad ohmic contacts, the difference in the state of the production technology
for each cell or the influence of intermediate layers [49].
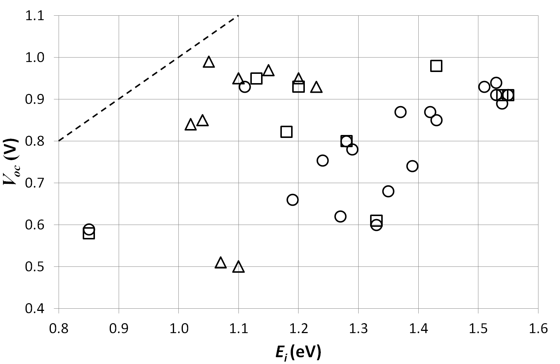
Figure 5. The open circuit voltage Voc of the studied solar cells versus the interface bandgap Ei. The circles and squares represent respectively cells where PC70BM and PC60BM are the acceptor material. The triangles represent cells with other acceptor materials. The dashed line indicates the limit for the Voc.
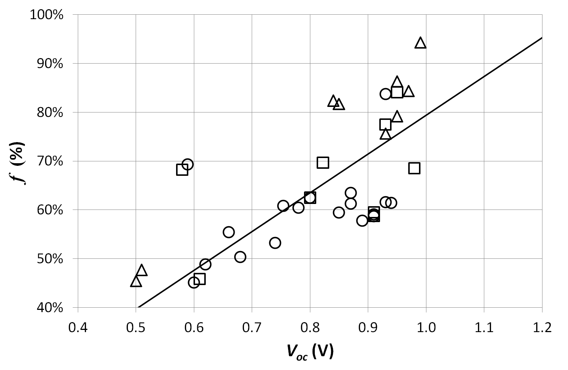
If we consider the interface bandgap Ei as the limit for the open circuit voltage Voc, the voltage factor f is given by f = (q. Voc / Ei) (equation 2).
Because of loss factors (e.g. recombination), f can not reach 1. Figure 6 shows a linear relationship between f and Voc, meaning that the higher the Voc, the more efficiently energy of the bandgap is used. These conclusions are the same as our 2006 study. The average voltage factor f increased from 53 % in 2006 to 65 % now (a relative increase of 23 %).
Finally, we compare the two most frequently used acceptors. Of the 36 solar cells, 18 cells use PC70BM as acceptor and 9 use PC60BM. The cells with PC70BM have an average efficiency of 5.9 %, with 9.2% as maximum. This is higher than the cells with PC60BM which have an average efficiency of 5.1 % (with 6.2 % as maximum). If we only consider the cells that use the same donor, but differ in acceptor, we find 5 pairs [10,16,19,20,23,25,28,31]. The cells with PC70BM as acceptor perform on average 21 % better than the same cells with PC60BM as acceptor (6.3 % average efficiency versus 5.2 %). The Voc and FF of both members of each pair don’t differ much. It is mainly the Jsc that improves by using PC70BM as acceptor. Taken into account that both acceptors have similar HOMO and LUMO energy levels, this difference can not be explained by the location of the energy levels. A possible explanation is the fact that PC60BM is a worse absorber than PC70BM, especially in the ultraviolet and visible light region [3].
Figure 6. The voltage factor f of the studied solar cells versus the open circuit voltage Voc. The circles and squares represent respectively cells where PC70BM and PC60BM are the acceptor material. The triangles represent cells with other acceptor materials. The straight line is the linear fit through the origin.
3. Conclusions
We compared the reported characteristics of 36 different bulk heterojunction organic solar cells fabricated and characterized by different research institutes. We confirmed the results from our 2006 study that not the Voc, but the Jsc is the limiting factor for efficiency improvements for the current generation of organic solar cells. Also, an empirical threshold of 0.3 eV was found between the LUMOs of donor and acceptor, necessary for exciton dissociation. A relation between the Voc and the interface bandgap was not found. The linear relationship between the voltage factor f and Voc indicates that the higher the Voc, the more efficiently energy of the bandgap is used. Finally, the cells in our study with PC70BM as acceptor perform significantly better than the same cells with PC60BM as acceptor.
References
1. Minnaert, B.; Burgelman, M., Empirical study of the characteristics of current-state organic bulk heterojunction solar cells. European Physical Journal-Applied Physics 2007, 38, 111-114.
2. Wang, X.; Chen, S.; Sun, Y.; Zhang, M.; Li, Y.; Li, X.; Wang, H., A furan-bridged D-pi-A copolymer with deep homo level: Synthesis and application in polymer solar cells. Polymer Chemistry 2011, 2, 2872-2877.
3. He, Y.; Li, Y., Fullerene derivative acc
eptors for high performance polymer solar cells. Physical Chemistry Chemical Physics 2011, 13, 1970-1983.
4. Schuenemann, C.; Wynands, D.; Wilde, L.; Hein, M.P.; Pfuetzner, S.; Elschner, C.; Eichhorn, K.-J.; Leo, K.; Riede, M., Phase separation analysis of bulk heterojunctions in small-molecule organic solar cells using zinc-phthalocyanine and C-60. Physical Review B 2012, 85.
5. Murtaza, I.; Qazi, I.; Karimov, K.S.; Sayyad, M.H., Direct current and impedance spectroscopic studies on MoO3 modified ZnPc/ITO schottky diodes. Physica B-Condensed Matter 2011, 406, 533-536.
6. Fan, B.; de Castro, F.A.; Heier, J.; Hany, R.; Nueesch, F., High performing doped cyanine bilayer solar cell. Organic Electronics 2010, 11, 583-588.
7. Chi, L.-C.; Chen, H.-F.; Hung, W.-Y.; Hsu, Y.-H.; Feng, P.-C.; Chou, S.-H.; Liu, Y.-H.; Wong, K.-T., Donor-acceptor small molecule with coplanar and rigid pi-bridge for efficient organic solar cells. Solar Energy Materials and Solar Cells 2013, 109, 33-39.
8. You, J.; Dou, L.; Yoshimura, K.; Kato, T.; Ohya, K.; Moriarty, T.; Emery, K.; Chen, C.-C.; Gao, J.; Li, G., et al., A polymer tandem solar cell with 10.6% power conversion efficiency. Nature Communications 2013, 4.
9. Zhang, G.; Fu, Y.; Zhang, Q.; Xie, Z., Benzo 1,2-b:4,5-b ' dithiophene-dioxopyrrolothiophen copolymers for high performance solar cells. Chemical Communications 2010, 46, 4997-4999.
10. Zhao, G.; He, Y.; Li, Y., 6.5% efficiency of polymer solar cells based on poly(3-hexylthiophene) and indene-C(60) bisadduct by device optimization. Advanced Materials 2010, 22, 4355-+.
11. Huang, C.-J.; Ke, J.-C.; Chen, W.-R.; Meen, T.-H.; Yang, C.-F., Improved the efficiency of small molecule organic solar cell by double anode buffer layers. Solar Energy Materials and Solar Cells 2011, 95, 3460-3464.
12. Lau, X.C.; Wang, Z.; Mitra, S., A C70-carbon nanotube complex for bulk heterojunction photovoltaic cells. Applied Physics Letters 2013, 103.
13. Aazou, S.; Ibral, A.; White, M.S.; Kaltenbrunner, M.; Glowacki, E.D.; Egbe, D.A.M.; Sariciftci, N.S.; Assaid, E.M., Organic bulk heterojunction solar cells based on P3HT and anthracene-containing PPE-PPV: Fabrication, characterization and modeling. Journal of Optoelectronics and Advanced Materials 2013, 15, 395-404.
14. Egbe, D.A.M.; Adam, G.; Pivrikas, A.; Ramil, A.M.; Birckner, E.; Cimrova, V.; Hoppe, H.; Sariciftci, N.S., Improvement in carrier mobility and photovoltaic performance through random distribution of segments of linear and branched side chains. Journal of Materials Chemistry 2010, 20, 9726-9734.
15. Chiu, S.W.; Lin, L.Y.; Lin, H.W.; Chen, Y.H.; Huang, Z.Y.; Lin, Y.T.; Lin, F.; Liu, Y.H.; Wong, K.T., A donor-acceptor-acceptor molecule for vacuum-processed organic solar cells with a power conversion efficiency of 6.4%. Chemical Communications 2012, 48, 1857-1859.
16. Yuan, Y.; Reece, T.J.; Sharma, P.; Poddar, S.; Ducharme, S.; Gruverman, A.; Yang, Y.; Huang, J., Efficiency enhancement in organic solar cells with ferroelectric polymers. Nature Materials 2011, 10, 296-302.
17. Zhou, J.; Wan, X.; Liu, Y.; Zuo, Y.; Li, Z.; He, G.; Long, G.; Ni, W.; Li, C.; Su, X., et al., Small molecules based on benzo 1,2-b:4,5-b ' dithiophene unit for high-performance solution-processed organic solar cells. Journal of the American Chemical Society 2012, 134, 16345-16351.
18. Liu, Y.; Larsen-Olsen, T.T.; Zhao, X.; Andreasen, B.; Sondergaard, R.R.; Helgesen, M.; Norrman, K.; Jorgensen, M.; Krebs, F.C.; Zhan, X., All polymer photovoltaics: From small inverted devices to large roll-to-roll coated and printed solar cells. Solar Energy Materials and Solar Cells 2013, 112, 157-162.
19. Chu, T.-Y.; Alem, S.; Verly, P.G.; Wakim, S.; Lu, J.; Tao, Y.; Beaupre, S.; Leclerc, M.; Belanger, F.; Desilets, D., et al., Highly efficient polycarbazole-based organic photovoltaic devices. Applied Physics Letters 2009, 95.
20. Chu, T.-Y.; Lu, J.; Beaupre, S.; Zhang, Y.; Pouliot, J.-R.; Zhou, J.; Najari, A.; Leclerc, M.; Tao, Y., Effects of the molecular weight and the side-chain length on the photovoltaic performance of dithienosilole/thienopyrrolodione copolymers. Advanced Functional Materials 2012, 22, 2345-2351.
21. Nie, W.; Coffin, R.C.; Liu, J.; Li, Y.; Peterson, E.D.; MacNeill, C.M.; Noftle, R.E.; Carroll, D.L., High efficiency organic solar cells with spray coated active layers comprised of a low band gap conjugated polymer. Applied Physics Letters 2012, 100.
22. Nie, W.; MacNeill, C.M.; Li, Y.; Noftle, R.E.; Carroll, D.L.; Coffin, R.C., A soluble high molecular weight copolymer of benzo 1,2-b:4,5-b ' dithiophene and benzoxadiazole for efficient organic photovoltaics. Macromolecular Rapid Communications 2011, 32, 1163-1168.
23. Wang, M.; Hu, X.; Liu, P.; Li, W.; Gong, X.; Huang, F.; Cao, Y., Donor acceptor conjugated polymer based on naphtho 1,2-c:5,6-c bis 1,2,5 thiadiazole for high-performance polymer solar cells. Journal of the American Chemical Society 2011, 133, 9638-9641.
24. Liu, Y.; Wan, X.; Wang, F.; Zhou, J.; Long, G.; Tian, J.; Chen, Y., High-performance solar cells using a solution-processed small molecule containing benzodithiophene unit. Advanced Materials 2011, 23, 5387-+.
25. Aich, B.R.; Beaupre, S.; Leclerc, M.; Tao, Y., Highly efficient thieno 3,4-c pyrrole-4,6-dione-based solar cells processed from non-chlorinated solvent. Organic Electronics 2014, 15, 543-548.
26. Yuan, M.-C.; Chou, Y.-J.; Chen, C.-M.; Hsu, C.-L.; Wei, K.-H., A crystalline low-bandgap polymer comprising dithienosilole and thieno 3,4-c pyrrole-4,6-dione units for bulk heterojunction solar cells. Polymer 2011, 52, 2792-2798.
27. Zhang, Y.; Zou, J.; Yip, H.-L.; Chen, K.-S.; Zeigler, D.F.; Sun, Y.; Jen, A.K.Y., Indacenodithiophene and quinoxaline-based conjugated polymers for highly efficient polymer solar cells. Chemistry of Materials 2011, 23, 2289-2291.
28. Li, W.; Furlan, A.; Hendriks, K.H.; Wienk, M.M.; Janssen, R.A.J., Efficient tandem and triple-junction polymer solar cells. Journal of the American Chemical Society 2013, 135, 5529-5532.
29. You, J.; Dou, L.; Hong, Z.; Li, G.; Yang, Y., Recent trends in polymer tandem solar cells research. Progress in Polymer Science 2013, 38, 1909-1928.
30. Dou, L.; You, J.; Yang, J.; Chen, C.-C.; He, Y.; Murase, S.; Moriarty, T.; Emery, K.; Li, G.; Yang, Y., Tandem polymer solar cells featuring a spectrally matched low-bandgap polymer. Nat. Photonics 2012, 6, 180-185.
31. Seo, J.H.; Gutacker, A.; Sun, Y.; Wu, H.; Huang, F.; Cao, Y.; Scherf, U.; Heeger, A.J.; Bazan, G.C., Improved high-efficiency organic solar cells via incorporation of a conjugated polyelectrolyte interlayer. Journal of the American Chemical Society 2011, 133, 8416-8419.
32. Chen, Y.-H.; Lin, L.-Y.; Lu, C.-W.; Lin, F.; Huang, Z.-Y.; Lin, H.-W.; Wang, P.-H.; Liu, Y.-H.; Wong, K.-T.; Wen, J., et al., Vacuum-deposited small-molecule organic solar cells with high power conversion efficiencies by judicious molecular design and device optimization. Journal of the American Chemical Society 2012, 134, 13616-13623.
33. Fan, X.; Cui, C.; Fang, G.; Wang, J.; Li, S.; Cheng, F.; Long, H.; Li, Y., Efficient polymer solar cells based on poly(3-hexylthiophene):Indene-C70 bisadduct with a MoO3 buffer layer. Advanced Functional Materials 2012, 22, 585-590.
34. He, Y.; Zhao, G.; Peng, B.; Li, Y., High-yield synthesis and electrochemical and photovoltaic properties of indene-c-70 bisadduct. Advanced Functional Materials 2010, 20, 3383-3389.
35. Sun, Y.; Welch, G.C.; Leong, W.L.; Takacs, C.J.; Bazan, G.C.; Heeger, A.J., Solution-processed small-molecule solar cells with 6.7% efficiency. Nature Materials 2012, 11, 44-48.
36. He, Z.; Zhong, C.; Su, S.; Xu, M.; Wu, H.; Cao, Y., Enhanced power-conversion efficiency in polymer solar cells using an inverted device structure. Nat. Photonics 2012, 6, 591-595.
37. Peumans, P.; Yakimov, A.; Forrest, S.R., Small molecular weight organic thin-film photodetectors and solar cells. Journal of Applied Physics 2003, 93, 3693-3723.
38. Scharber, M.C.; Wuhlbacher, D.; Koppe, M.; Denk, P.; Waldauf, C.; Heeger, A.J.; Brabec, C.L., Design rules for donors in bulk-heterojunction solar cells - towards 10 % energy-conversion efficiency. Advanced Materials 2006, 18, 789-+.
39. Lunt, R.R.; Osedach, T.P.; Brown, P.R.; Rowehl, J.A.; Bulovic, V., Practical roadmap and limits to nanostructured photovoltaics. Advanced Materials 2011, 23, 5712-5727.
40. Tadayyon, S.; Grandin, H.; Griffiths, K.; Coatsworth, L.; Norton, P.; Aziz, H.; Popovic, Z., Reliable and reproducible determination of work function and ionization potentials of layers and surfaces relevant to organic light emitting diodes. Organic electronics 2004, 5, 199-205.
41. Djurovich, P.I.; Mayo, E.I.; Forrest, S.R.; Thompson, M.E., Measurement of the lowest unoccupied molecular orbital energies of molecular organic semiconductors. Organic Electronics 2009, 10, 515-520.
42. Dennler, G.; Sariciftci, N.S., Flexible conjugated polymer-based plastic solar cells: From basics to applications. Proceedings of the Ieee 2005, 93, 1429-1439.
43. Hoppe, H.; Egbe, D.A.M.; Muhlbacher, D.; Sariciftci, N.S., Photovoltaic action of conjugated polymer/fullerene bulk heterojunction solar cells using novel ppe-ppv copolymers. Journal of Materials Chemistry 2004, 14, 3462-3467.
44. Gadisa, A.; Svensson, M.; Andersson, M.R.; Inganas, O., Correlation between oxidation potential and open-circuit voltage of composite solar cells based on blends of polythiophenes/fullerene derivative. Applied Physics Letters 2004, 84, 1609-1611.
45. Kooistra, F.B.; Knol, J.; Kastenberg, F.; Popescu, L.M.; Verhees, W.J.H.; Kroon, J.M.; Hummelen, J.C., Increasing the open circuit voltage of bulk-heterojunction solar cells by raising the lumo level of the acceptor. Organic Letters 2007, 9, 551-554.
46. Brabec, C.J.; Cravino, A.; Meissner, D.; Sariciftci, N.S.; Fromherz, T.; Rispens, M.T.; Sanchez, L.; Hummelen, J.C., Origin of the open circuit voltage of plastic solar cells. Advanced Functional Materials 2001, 11, 374-380.
47. Morvillo, P.; Bobeico, E., Tuning the lumo level of the acceptor to increase the open-circuit voltage of polymer-fullerene solar cells: A quantum chemical study. Solar Energy Materials and Solar Cells 2008, 92, 1192-1198.
48. Mihailetchi, V.D.; Blom, P.W.M.; Hummelen, J.C.; Rispens, M.T., Cathode dependence of the open-circuit voltage of polymer : Fullerene bulk heterojunction solar cells. Journal of Applied Physics 2003, 94, 6849-6854.
49. Liu, Z.; Lo, M.; Wang, H.; Ng, T.; Roy, V.; Lee, C.; Lee, S., Influence of the donor/acceptor interface on the open-circuit voltage in organic solar cells. Applied Physics Letters 2009, 95, 093307-093307-093303.